CANCER
Treatment advances in metastatic lung cancer
An overview of treatment advances in metastatic lung cancer
December 16, 2015
-
Lung cancer is the leading cause of cancer-related mortality worldwide. Non-small cell lung cancer (NSCLC) accounts for approximately 80-85% of all lung cancers. Despite increased awareness and improved treatment techniques, the majority of patients present with locally advanced or metastatic disease at diagnosis. The median survival rarely exceeds 10 to 12 months in unselected patients with metastatic disease treated with conventional chemotherapy.
Targeted agents
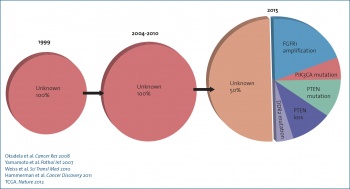
Advances in molecular genotyping in recent years, as well as a better understanding of the concept of ‘oncogene addiction’, have led to the development of targeted agents with improved survival outcomes in subsets of patients with NSCLC, especially adenocarcinomas and non-smokers (Figure 1). The most notable examples are the epidermal growth factor receptor (EGFR) activating mutations and the echinoderm microtubule-associated protein-like 4-anaplastic lymphoma kinase (EML4-ALK) translocation. Although only 12-15% of NSCLC tumours are EGFR-mutated or ALK-rearrangement-positive,1 this translates into a considerable number of patients affected worldwide.
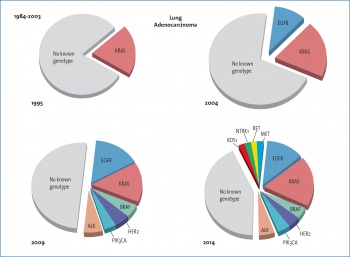 Figure 1. Lung cancer therapy 2015 – Identification of genomic alterations for NSCLC(click to enlarge)
Figure 1. Lung cancer therapy 2015 – Identification of genomic alterations for NSCLC(click to enlarge)In metastatic NSCLC, presence of EGFR activating mutations confers a more favourable prognosis and strongly predicts for sensitivity to EGFR tyrosine kinase inhibitors (TKIs), such as gefitinib, erlotinib or afatinib.2 A number of large randomised trials comparing an EGFR-TKI v. standard platinum-based chemotherapy have clearly demonstrated the superiority of the target agent in prolonging progression-free survival (PFS), improving response rate (RR) along with a more favourable safety profile.3 Similarly, in the presence of an EML4-ALK translocation, the ALK inhibitor crizotinib produces higher RR and longer PFS when compared to standard chemotherapy,4 both in the first- and second-line settings.
Beyond EGFR mutations and ALK translocation, novel biomarkers have been linked with adenocarcinoma histology, including ROS1 fusion gene, MET amplification or mutations, BRAF and HER2 mutations, for which many targeted drugs are currently under investigation. ROS1 re-arrangements and MET amplification represent two additional molecular aberrations that predict for crizotinib sensitivity.5,6 Results from expansion cohorts of the PROFILE 1001 phase 1 trial suggested the potential efficacy of crizotinib in lung cancer patients with ROS1 translocation or MET amplification. BRAF mutations are another promising target, occurring in 1% to 3% of adenocarcinoma patients, including those with a history of smoking. Recently, a combination of the BRAF inhibitor dabrafenib with the MEK inhibitor trametinib emerged as a suitable strategy in patients with BRAF mutant NSCLC. Dual BRAF/MEK axis inhibition has been shown to translate into a RR of 63% with an acceptable toxicity profile.7
Squamous cell carcinoma
Squamous cell carcinoma (SCC) of the lung accounts for 20-30% of all non-small cell lung cancers (NSCLC). Compared to adenocarcinomas, advances in lung SCC have been far more modest. At present, there are no targeted agents approved for the treatment of lung SCC (Figure 2) however, the last year has seen an unprecedented evolution in the management of lung SCC, with survival gains noted in both the first- and second-line setting in randomised clinical trials. Unfortunately, to date, the identification of oncogenic driver mutations in lung SCC has yet to yield the significant improvements seen in non-squamous histology.
In the first-line setting, the addition of the second-generation recombinant human IgG1 EGFR monoclonal antibody (Mab), necitumumab, to gemcitabine and cisplatin has been shown to improve overall survival (OS) by 1.6 months (11.5 months v. 9.9 months) in the phase 3 open-label SQUIRE trial, with comparable adverse events.8 Most recently, the results of the phase 3 LUX-Lung 8 trial of afatinib v. erlotinib in patients with lung SCC progressing after four cycles of platinum-based chemotherapy have been published, demonstrating improved OS with afatinib 7.9 months v. 6.8 months, with similar adverse events profiles noted between the two groups.9
The inhibition of angiogenesis has proven challenging in the treatment of patients with lung SCC due to the central location of these tumours and their close proximity to large blood vessels in the chest wall. Findings from newer second-generation angiogenesis inhibitors, however, show comparable levels of gastrointestinal and respiratory tract bleeding events across all NSCLC histologies. Compared to placebo, the anti-VEGFR-2 IgG MAb, ramucirumab, has recently been shown to improve PFS (4.5 months v. 3 months) and OS (10.5 months v. 9.1 months) in patients with advanced NSCLC progressing after first-line platinum-based chemotherapy. Significant improvements have also been demonstrated in patients with lung SCC in terms of overall objective response (26.8% v. 10.5%), disease control rate (59.9% v. 45%) and PFS (4.2 months v. 2.7 months) and an OS benefit (9.5 months v. 8.2 months). In Dec 2014, ramucirumab received FDA approval for use with docetaxel in the second-line management of advanced NSCLC, including patients with squamous histology.10
The inhibition of T-cell activation through programmed death (PD-1) receptor interaction with the tumour expressing PD-L1 ligand (immune checkpoint) is a noted mechanism of tumour immune surveillance escape in NSCLC. In the management of lung SCC, the fully human IgG4 anti-PD-1 monoclonal antibody, nivolumab, has recently replaced docetaxel as the preferred second-line therapy based on the results of CHECKMATE 017, a phase 3 study of nivolumab v. docetaxel.
Findings in CHECKMATE 017 demonstrated improved median OS of 3.2 months (9.2 months v. six months) and improved one-year survival over docetaxel (42% v. 24%), with a more favourable safety profile and fewer treatment related grade 3/4 AE (7% v. 55%).11 With the recent FDA approval of nivolumab in the second-line setting in March 2015, docetaxel will likely be relegated to third-line therapy in the management of lung SCC. However, additional studies are required to confirm the results of CHECKMATE 017 given the lower than expected median survival observed in the docetaxel arm, to identify biomarkers of response, and to better define the unique toxicities associated with these immune-modulating agents (Table 1).
Lung-MAP
The Lung-MAP project is a large biomarker-driven clinical trial in patients with lung SCC, designed to facilitate approval of targeted therapy-predictive biomarker combinations.12 In Lung-MAP, each patient’s tumour tissue is analysed for more than 200 genomic alterations. The results of this analysis are then used to offer these patients participation in one of five phase 2 or 3 randomised trials, or substudies, within the Lung-MAP framework.
Overall, four of the Lung-MAP substudies test new targeted therapies. For example, in one study, open to patients whose tumours harbour PIK3CA gene mutations, patients are randomly assigned to treatment with the PI3K inhibitor GDC-0032 or docetaxel. In a study open to patients whose tumours demonstrate FGFR1, FGFR2, or FGFR3 gene amplification or mutations, patients are randomly assigned to treatment with the FGFR inhibitor AZD4547 or docetaxel. Patients without genetic alterations that match one of the tested targeted therapies are offered a randomised trial in which patients receive immunotherapy or docetaxel (Figure 3).
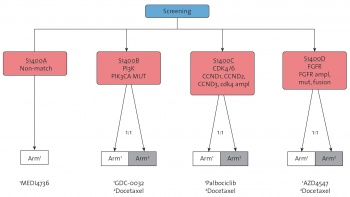 Figure 3. Lung-MAP: Patients are assigned to the trial arm that is best matched to their tumour's genomic profile(click to enlarge)
Figure 3. Lung-MAP: Patients are assigned to the trial arm that is best matched to their tumour's genomic profile(click to enlarge)Personalised medicine
Unlike ‘basket trials’, where researchers test the effect of a single drug on a single mutation in a variety of cancer types, ‘umbrella’ trials are designed to test the impact of personalised medicine with different drugs on different mutations in a single type of cancer. The phase 2 BATTLE (Biomarker-integrated Approaches of Targeted Therapy for lung Cancer) trial was the first prospective, biopsy-mandated, biomarker-based study that adaptively randomised 255 pre-treated NSCLC patients to erlotinib, sorafenib, erlotinib plus bexarotene, or vandetanib, based on molecular biomarker analysed in fresh core needle biopsy specimens.13
Overall results included a 46% eight-week disease control rate which was the primary endpoint. This trial established the feasibility of ‘real-time’ biopsies and personalised treatment in lung cancer. BATTLE-2 (NCT01248247), a phase 2, randomised, multi-arm study in advanced pre-treated EGFR wild-type and ALK non-rearranged NSCLC patients is currently ongoing.14
New era in lung cancer research
Intratumoural heterogeneity plays a critical role in tumour evolution. Molecular characterisation of the tumour is typically provided from a single biopsy and at single time point. Multiregional evaluations to determine geographical heterogeneity, and molecular characterisation of different samples collected over space and time to ascertain clonal evolution are not routinely carried out.
The prospective TRACERx trial (TRAcking non-small cell lung Cancer Evolution through therapy [Rx]) in NSCLC patients, aims to define the evolutionary trajectories of lung cancer in both space and time through multiregion and longitudinal tumour sampling and genetic analysis by following cancer from diagnosis to relapse.15
In the near future most patients with metastatic tumours will receive targeted therapies or immune modulators delineated by tumour genotyping and analysis of immune contexture and all of these trials will help to validate current biomarkers facilitating rapid access to innovative therapies.
References
- Yang Jin-Ji, et al. Lung cancers with concomitant EGFR mutations and ALK rearrangements: diverse responses to EGFR-TKI and crizotinib in relation to diverse receptors phosphorylation. Clinical Cancer Research 2014: 20(5); 1383-1392
- Domvri Kalliopi, et al. Molecular targeted drugs and biomarkers in NSCLC, the evolving role of individualized therapy. Journal of Cancer 2013; 4(9): 736
- Giovannetti Elisa, et al. Association of polymorphisms in AKT1 and EGFR with clinical outcome and toxicity in Non–Small Cell Lung Cancer patients treated with gefitinib. Molecular cancer therapeutics 2010; 19(3): 581-593
- Costa Daniel B, et al. Clinical experience with crizotinib in patients with advanced ALK-rearranged non–small-cell lung cancer and brain metastases. Journal of Clinical Oncology 2015; doi: 10.1200/JCO.2014.59.0539
- Bergethon Kristin, et al. ROS1 rearrangements define a unique molecular class of lung cancers. Journal of clinical oncology 2012; 30(8): 863-870
- Lennerz Jochen K, et al. MET amplification identifies a small and aggressive subgroup of esophagogastric adenocarcinoma with evidence of responsiveness to crizotinib. Journal of Clinical Oncology 2011; 29(36): 4803-4810
- Joshi Monika, et al. Trametinib with or without Vemurafenib in BRAF Mutated Non-Small Cell Lung Cancer. PloS one 2015; 12(2): e0118210
- Thatcher Nick, et al. Necitumumab plus gemcitabine and cisplatin v. gemcitabine and cisplatin alone as first-line therapy in patients with stage IV squamous non-small-cell lung cancer (SQUIRE): an open-label, randomised, controlled phase 3 trial. The Lancet Oncology 2015; 16(7): 763-774
- Soria Jean-Charles, et al. Afatinib v. erlotinib as second-line treatment of patients with advanced squamous cell carcinoma of the lung (LUX-Lung 8): an open-label randomised controlled phase 3 trial. The Lancet Oncology 2015; 16(8): 897-907.
- Garon Edward B, et al. Ramucirumab plus docetaxel v. placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. The Lancet 2014; 384(9944): 665-673
- Brahmer Julie, et al. Nivolumab v. docetaxel in advanced squamous-cell non–small-cell lung cancer. New England Journal of Medicine 2015; 373(2): 123-135
- Herbst Roy S, et al. Lung Master Protocol (Lung-MAP) - a biomarker-driven protocol for accelerating development of therapies for squamous cell lung cancer: SWOG S1400. Clinical Cancer Research 2015; 21(7): 1514-1524
- Kim Edward S, et al. The BATTLE trial: personalizing therapy for lung cancer. Cancer discovery 2011; 1(1): 44-53
- Papadimitrakopoulou Vassiliki, et al. BATTLE-2 program: a biomarker-integrated targeted therapy study in previously treated patients with advanced non-small cell lung cancer (NSCLC). Journal of Thoracic Oncology 2013; 8: 530
- Jamal-Hanjani Mariam, et al. Tracking genomic cancer evolution for precision medicine: the lung TRACERx study 2014; e1001906