HEALTH SERVICES
MENTAL HEALTH
Carbamazepine therapy in a long-stay ward for patients with intellectual disability
A 12-month retrospective audit of patients on carbamazepine in a long-stay residential ward
April 12, 2018
-
In a long-stay residential ward for patients with intellectual disability it was noticed that a large proportion of patients had hyponatraemia when their renal profile work-up was done as part of scheduled reviews. All the patients who had hyponatraemia were on carbamazepine and three of them were on a combination of a serotonin reuptake inhibitor (SSRI) and carbamazepine. One patient was severely symptomatic and was admitted with hyponatraemia related seizures to the Midland Regional Hospital, Portlaoise.
There were 28 patients – 46.4% (13) females and 53.6% (15) males – in our study group.
Carbamazepine is a commonly prescribed medication in psychiatry and produces side effects in 33-50% of patient.1
Carbamazepine was prescribed for either problem behaviour and/or epilepsy.
The audit
Serum sodium level in the 12 months preceding the time of audit and the most recent serum sodium levels were checked for the 28 residents; the mean serum sodium levels were 137.7mmol/L (preceding audit) and 135.1mmol/L (most recent level). Hyponatraemia was taken as a serum sodium level of <135mmol/L and all blood tests were carried out in the biochemistry laboratory at the hospital.
Results
36% (10) of the residents were between 30-50 years old and 64% (18) were > 50 years old
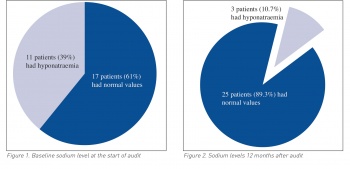
39% (11) of residents had hyponatraemia at the start of the audit (in the preceding 12 months) and 61% (17) had normal values. Out of 11 patients who had hyponatraemia, 63.6% (7) were females and 36.4% (4) were males
The age range of the patients who had hyponatraemia was 42-73 years (mean age = 61.5 years), while the age range of patients with normal values was 38-69 years (mean age 56 years)
35.3% (6) of those with normal values were females and 64.7% (11) were males
Of the 11 patients who had hyponatraemia at the start of the audit (preceding 12 months), 72.7% (8) were on carbamazepine alone and 27.3% (3) were on both carbamazepine and SSRI. The mean serum sodium level was 130.7mmol/L, mean carbamazepine dose was 412mg/day and mean trough level was 10.7ug/ml (8-12ug/ml)
None of the patients on carbamazepine had normal values; 17.8% (5) patients on SSRIs had normal sodium levels with a mean serum sodium level of 138.3mmol/L.
We started reducing the dose of patients’ carbamazepine and also advised general practitioners who prescribed carbamazepine for patients with epilepsy to consider a change of medication as hyponatraemia is a known side effect of carbamazepine therapy.
12 months after audit
Eight patients remained on carbamazepine at reduced dose, five patients were still on SSRIs and three patients remained on both carbamazepine and SSRIs.
10.7% (3) of patients in total had hyponatraemia, 7.1% (2) on carbamazepine and 3.6% (1) of patients on both carbamazepine as well as SSRI; 17.9% (5) of patients on SSRI had normal values.
Findings
Hyponatraemia was found to be more common in women
Hyponatraemia was associated with higher mean age compared to patients with normal values
There was a good response overall to reduction in dose of carbamazepine resulting in normonatraemia.
Discussion
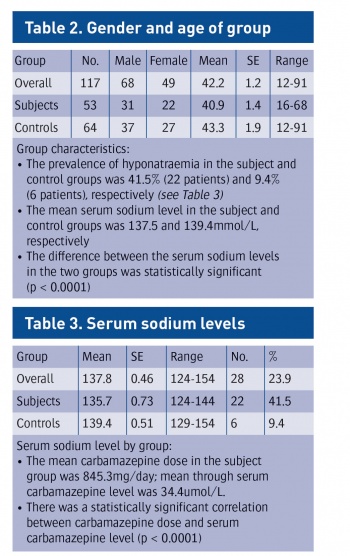
A retrospective chart review of all residents at a residential centre for people with intellectual disability (ID) was undertaken. All people receiving carbamazepine were identified, and the most recent serum sodium and carbamazepine levels were ascertained. All the people who were receiving carbamazepine, and had recorded serum sodium and carbamazepine levels from the previous two years were included as subjects (n = 53).
All people who were not receiving carbamazepine and had recorded serum sodium levels from the previous two years were included as controls (n =64).
Hyponatraemia was defined as a serum sodium level of < 135mmol/L. This is the value that has been used in comparable previous studies.2,3
All blood tests were carried out at the same laboratory, which also defines 135mmol/L as the lower limit of the normal range for serum sodium.
The carbamazepine blood levels were trough levels taken 12 hour after the administration of carbamazepine.
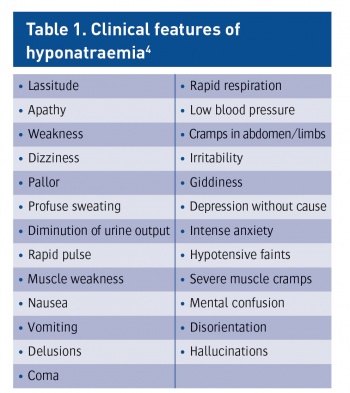
A checklist of symptoms of hyponatraemia was developed, based on clinical descriptions of the condition.4
A single investigator assessed all the subjects and controls through clinical observation and interviews with nursing staff. The checklist consisted of 25 items (see Table 1).
Serum sodium levels were compared between the subject and control groups; the relationships between serum sodium level, sex, age, daily carbamazepine dose and serum carbamazepine level were also examined. There was no significant difference between the subject and control groups in terms of age or sex (see Table 2) and males outnumbered females in both groups. The mean age of the participants in the study was 42.18 years (range = 12-91 years).
 (click to enlarge)
(click to enlarge)

 (click to enlarge)
(click to enlarge)
 (click to enlarge)
(click to enlarge)